This web page was produced as an assignment for Gen677 at UW-Madison Spring 2013
Protein Domains of MYO5a
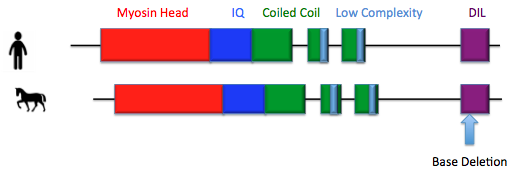
There are four different protein domains in MYO5a, a myosin head domain, an IQ domain, a coiled coil domain and a DIL domain[1]. The myosin head domain is the portion of the protein that binds to actin and also contains the catalytic center of the protein where ATP is turned into motive force allowing MYO5a to move along the actin filament[1]. The IQ domain is a set of six binding sites where myosin light chains and regulatory proteins can bind[1]. The coiled coil domain is the part the protein and bind to and interacts with another copy of MYO5a allowing the protein to dimerize[1]. Finally, the DIL domain is the binding site of the different cargos and MYO5a transports around the cell[1]. It is in this DIL domain that the mutation causing lavender foal syndrome, resulting in a nonfunctional protein and is unable to bind to cellular cargo[2].
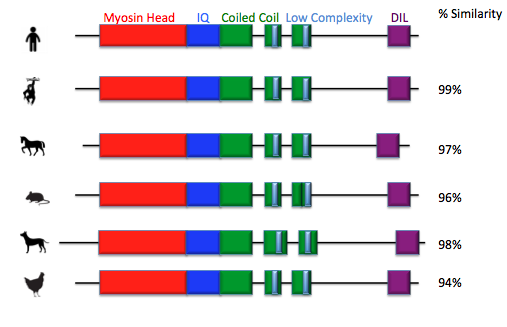
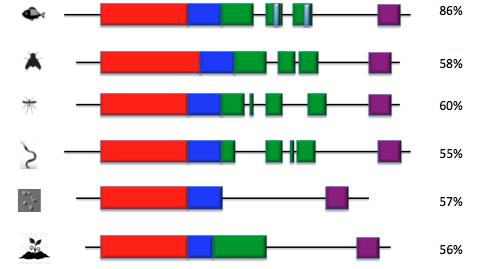
The four domain are highly conserved across all species from human to arabadopsis.
Analysis
There is a set of low complexity regions that are highly conserved in vertebrates. A low complexity region is a part of the protein that is highly conserved and contains little diversity [3]. The regions are found in the coiled coil domain. These regions could be an uncharacterized domain in MYO5a but no function has as yet been assigned to them [3]. This provides an interestion topic for future research into the function of MYO5a.
References
[1] Desnos C, Huet S, Darchen F. (2007). "Should I stay or should I go?": myosin V function in organelle trafficking. Biol Cell 99:411-423.
[2] Brooks SA, Gabreski N, Miller D, Brisbin A, Brown HE, et al. (2010) Whole-Genome SNP Association in the Horse: Identification of a Deletion in Myosin VA Responsible for Lavender Foal Syndrome. PLoS Genet 6(4): e1000909. doi:10.1371/journal.pgen.1000909.
[3] Coletta A, Pinney JW, Solis DYW, Marsh J, Pettifer SR, Attwood TK. (2010). Low-complexity regions within protein sequences have position-dependent roles. BMC Systems Biology 4:43
[2] Brooks SA, Gabreski N, Miller D, Brisbin A, Brown HE, et al. (2010) Whole-Genome SNP Association in the Horse: Identification of a Deletion in Myosin VA Responsible for Lavender Foal Syndrome. PLoS Genet 6(4): e1000909. doi:10.1371/journal.pgen.1000909.
[3] Coletta A, Pinney JW, Solis DYW, Marsh J, Pettifer SR, Attwood TK. (2010). Low-complexity regions within protein sequences have position-dependent roles. BMC Systems Biology 4:43
Rebecca Blackburn, [email protected], 5/16/13